In May, the OpenTrialsFDA team (a collaboration between Erick Turner, Dr. Ben Goldacre and the OpenTrials team at Open Knowledge) was selected as a finalist for the Open Science Prize. This global science competition is focused on making both the outputs from science and the research process broadly accessible to the public. Six finalists will present their final prototypes at an Open Science Prize Showcase in early December 2016, with the ultimate winner to be announced in late February or early March 2017.
 As the name suggests, OpenTrialsFDA is closely related to OpenTrials, a project funded by The Laura and John Arnold Foundation that is developing an open, online database of information about the world’s clinical research trials. OpenTrialsFDA will work on increasing access, discoverability and opportunities for re-use of a large volume of high quality information currently hidden in user-unfriendly Food and Drug Administration (FDA) drug approval packages (DAPs).
As the name suggests, OpenTrialsFDA is closely related to OpenTrials, a project funded by The Laura and John Arnold Foundation that is developing an open, online database of information about the world’s clinical research trials. OpenTrialsFDA will work on increasing access, discoverability and opportunities for re-use of a large volume of high quality information currently hidden in user-unfriendly Food and Drug Administration (FDA) drug approval packages (DAPs).
The FDA publishes these DAPs as part of the general information on drugs via its data portal known as Drugs@FDA. These documents contain detailed information about the methods and results of clinical trials, and are unbiased, compared to reports of clinical trials in academic journals. This is because FDA reviewers require adherence to the outcomes and analytic methods prespecified in the original trial protocols, so, in contrast to most journal editors, they are unforgiving of practices such as post hoc switching of outcomes and changes to the planned statistical analyses. These review packages also often report on clinical trials that have never been published.
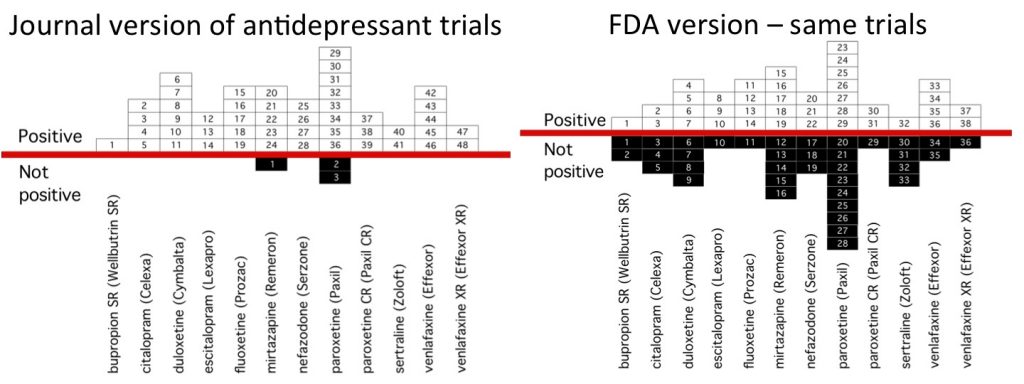
However, despite their high value, these FDA documents are notoriously difficult to access, aggregate, and search. The website itself is not easy to navigate, and much of the information is stored in PDFs or non-searchable image files for older drugs. As a consequence, they are rarely used by clinicians and researchers. OpenTrialsFDA will work on improving this situation, so that valuable information that is currently hidden away can be discovered, presented, and used to properly inform evidence-based treatment decisions.
The team has started to scrape the FDA website, extracting the relevant information from the PDFs through a process of OCR (optical character recognition). A new OpenTrialsFDA interface will be developed to explore and discover the FDA data, with application programming interfaces (APIs) allowing third party platforms to access, search, and present the information, thus maximising discoverability, impact, and interoperability. In addition, the information will be integrated into the OpenTrials database, so that for any trial for which a match exists, users can see the corresponding FDA data.
Future progress will be shared both through this blog and the OpenTrials blog and through fda.opentrials.net: you can also sign up for the OpenTrials newsletter to receive regular updates and news. More information about the Open Science Prize and the other finalists is available from www.openscienceprize.org/res/p/finalists.
Contact: opentrials@okfn.org
Twitter: @opentrials
As Communications Officer, Lieke works on increasing the profile and awareness of Open Knowledge Foundation projects online. She previously coordinated the OpenGLAM initiative, promoting free and open access to digital cultural heritage data and has been managing European projects in the areas of open cultural data, open access and open science. She is based in Berlin, where she also serves as Community Director of the Disruption Network Lab.